Exam-Style Problem
⬅ Back to Subchapter
Browsing as Guest. Progress, bookmarks and attempts are disabled.
Log in to track your work.
June 2017 p33 q8
2322
In a certain chemical reaction, a compound A is formed from a compound B. The masses of A and B at time t after the start of the reaction are x and y respectively and the sum of the masses is equal to 50 throughout the reaction. At any time the rate of increase of the mass of A is proportional to the mass of B at that time.
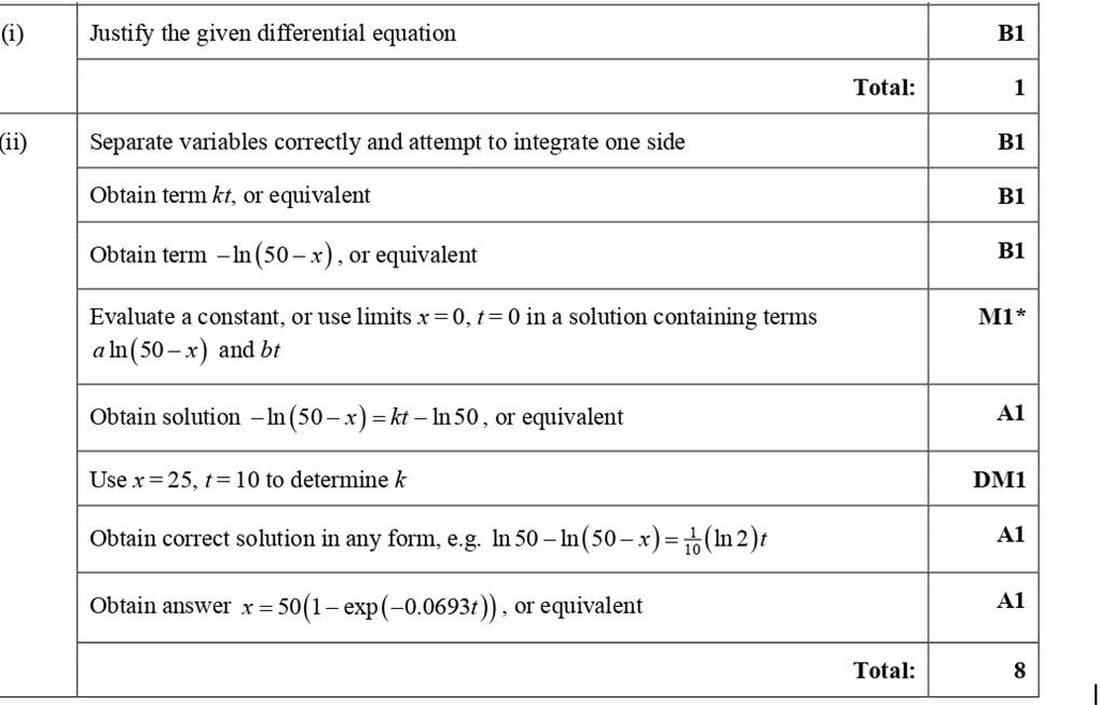
(i) Explain why \(\frac{dx}{dt} = k(50 - x)\), where k is a constant.
It is given that \(x = 0\) when \(t = 0\), and \(x = 25\) when \(t = 10\).
(ii) Solve the differential equation in part (i) and express x in terms of t.
Log in to record attempts.